320 and 130 percent error (%)
Percentage (%) Error Calculator
How to calculate Percent Error - Step by Step Solution
The Percent Calculator (Error) uses this formula:
The terms "experimental" and "theoretical" used in the equation above are commonly replaced with similar terms. Some terms also used for experimental are "measured," or "actual" and for theoretical the terms "accepted" and "calculated" are frequently used.
The Percent Error can be a positive or a negative number. If the experimental value is greater than the theoretical value, the result will be positive. On the other hand, if the experimental value is smaller than the theoretical value, the result will be negative. See more about percent error here.
This calculator can answers to questions like:
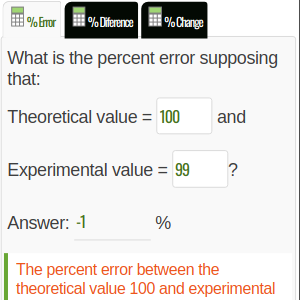
1) What is the percent error supposing 130 as the measured value and 320 as the calculated value?
2) What is the percent error supposing 320 as the measured value and 130 as the calculated value?
3) What is the absolute error between 320 and 130 supposing 320 as the theoretical value? Or may be: 320 and 130 percent error (%)
See the solutions to these problems below.
1) What is the percent error supposing 130 as the measured value and 320 as the calculated value?
Use the formula above to find the percent error. So, replacing the given values, we have
Percent error = [(130 - 320) / 320] x 100
= (-190 / 320) x 100
= -59.375 % (negative error)
In this case we have a % of negative error because the experimental is smaller than the theoretical.
2) What is the absolute error between 320 and 130 considering 320 as the theoretical value?
This problem is not about percent or relative error, but about absolute error. The solution is very simple:
= 130 - 320 = -190 (negative error)
Percentage (%) Error Calculator
Please link to this page! Just right click on the image below, choose copy link address, then past it in your HTML or in your social media app.